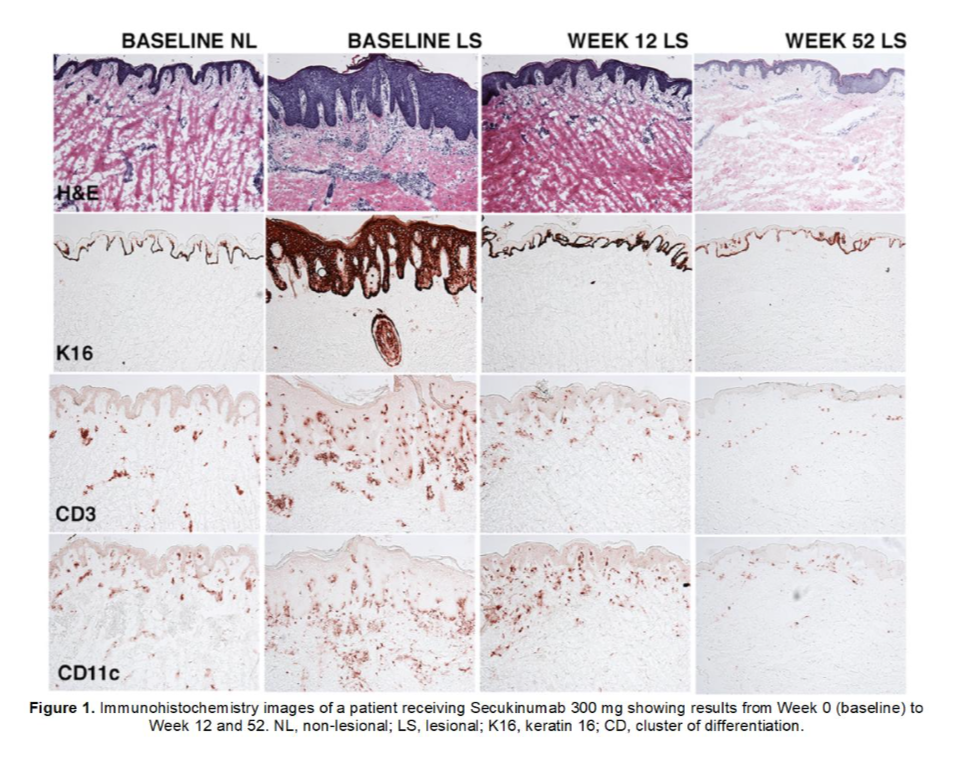
Keratin 16 (K16) is a marker of keratinocyte hyperproliferation, a process that plays a pivotal role in the pathogenesis of psoriasis. In patients with psoriasis, there is overexpression of K16 which correlates with overproduction of keratinocyte-derived pro-inflammatory molecules.
In the ObePso-S study, normalization of K16 was observed in patients receiving Secukinumab (Scapho®). In just 12 weeks, biopsy evaluation showed that 79.6% of patients receiving Secukinumab (Scapho®) 300 mg had no detectable K16 expression (Fig. 1).
Furthermore, 55.8% of patients receiving Secukinumab (Scapho®) achieved Psoriasis Area and Severity Index (PASI) 90 at Week 12, which increased to 59.6% at Week 52. This shows that sustained clinical response with Secukinumab (Scapho®) was associated with normalization of K16 in patients with psoriasis.
This study demonstrated that early reversal of histologic changes associated with treatment of Secukinumab (Scapho®) was associated with long-term successful disease control and confirmed the known clinical response in patients with moderate-to-severe psoriasis.
The ObePso-S study is a randomized, double-blind, placebo-controlled, phase IV trial conducted over 52 weeks. Patients were given either Secukinumab (Scapho®) 300 mg (N=54) or placebo (N=28) at Weeks 0, 1, 2, 3, 4, and monthly thereafter.
For more information on Secukinumab (Scapho®), read the full prescribing information here.
Reference: Blauvelt et al. (2023). ObePso-S Study. Journal of Dermatological Science. 109:12-21.