If we can aim for 100% skin clearance, then let’s go for it!
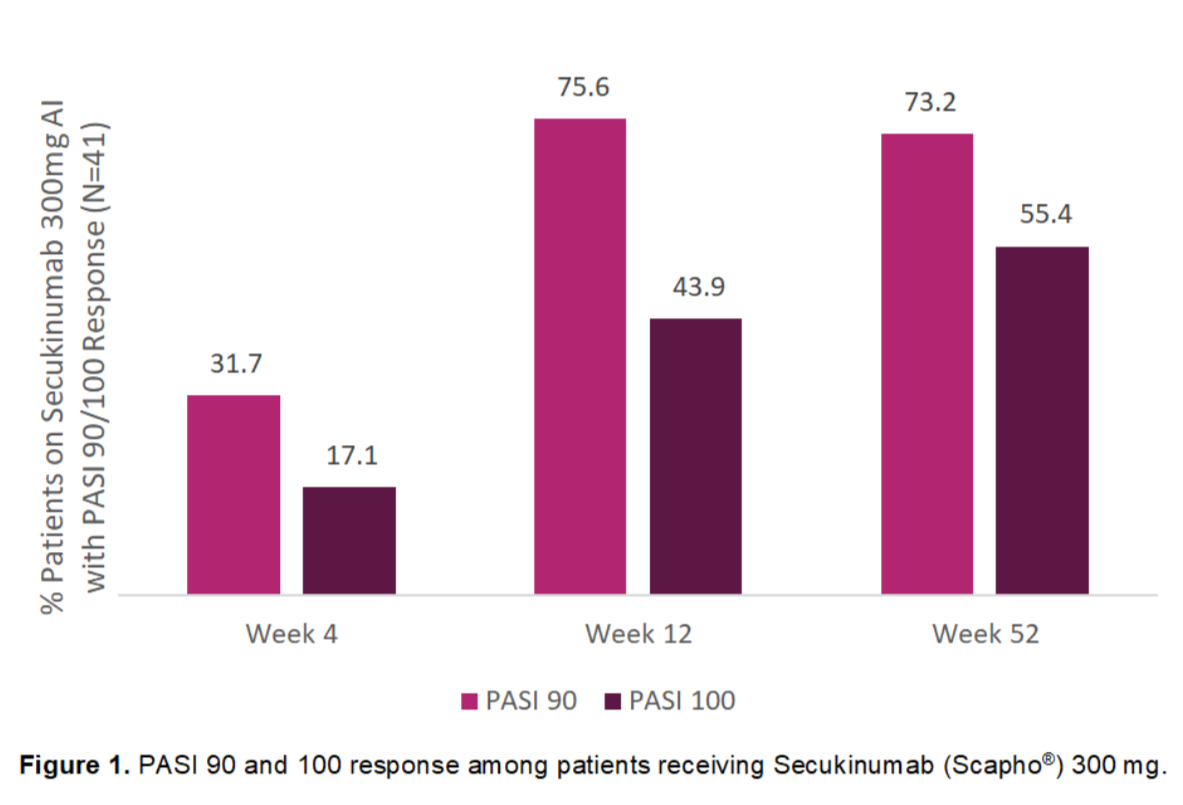
In the MATURE study, Psoriasis Area and Severity Index (PASI) 100 has been observed as early as in Week 4 in 17.1% of patients receiving Secukinumab (Scapho®) 300 mg. This number further improved as more patients achieved 100% skin clearance at Weeks 12 and 52 (Fig. 1).
Furthermore, 31.7% of patients receiving Secukinumab (Scapho®) 300 mg achieved PASI 90 at Week 4 which increased to 75.6% at Week 12 and 73.2% at Week 52 (Fig. 1).
This study also showed that patients have a high degree of satisfaction with the use of the autoinjector format versus the pre-filled syringe, independently of whether it was containing Secukinumab (Scapho®) or placebo. Moreover, no new safety signals were identied.
This study demonstrated that patients on Secukinumab (Scapho®) can achieve almost clear to clear skin as early as after the initiation phase, with the comfort of a convenient device.
The MATURE study is a randomized, double-blind, placebo-controlled, phase III trial conducted over 52 weeks. Patients were given either Secukinumab (Scapho®) 300 mg autoinjector (N=41), pre-filled syringe (N=41), or placebo (N=40) at Weeks 0, 1, 2, 3, 4, and monthly thereafter.
For more information on Secukinumab (Scapho®), read the full prescribing information here.
Reference: Sigurgeirsson et al. (2022). MATURE Study. Dermatologic Therapy. 35:e15285.