Use Ruxolitinib (JAKAVI®) as soon as MF patients are eligible to bring the most benefit to their lives 1-4
Patients with MF face increasingly debilitating symptoms as the disease progresses, with symptoms that negatively affect quality of life (QoL)5-6
Ruxolitinib (JAKAVI) can change lives in MF by offering patients rapid and durable improvements in MF-related symptoms and splenomegaly, regardless of MF risk status7-8
Using Ruxolitinib (JAKAVI) early may increase benefits for eligible patients with MF, helping to improve their symptom burden so they can get back to being more like themselves 9-10
An ad-hoc analysis of JUMP§ showed that low-risk patients achieved slightly greater spleen size reductions vs high-risk patients, suggesting that earlier Ruxolitinib (JAKAVI) treatment may lead to greater benefits in patients with myelofibrosis.10
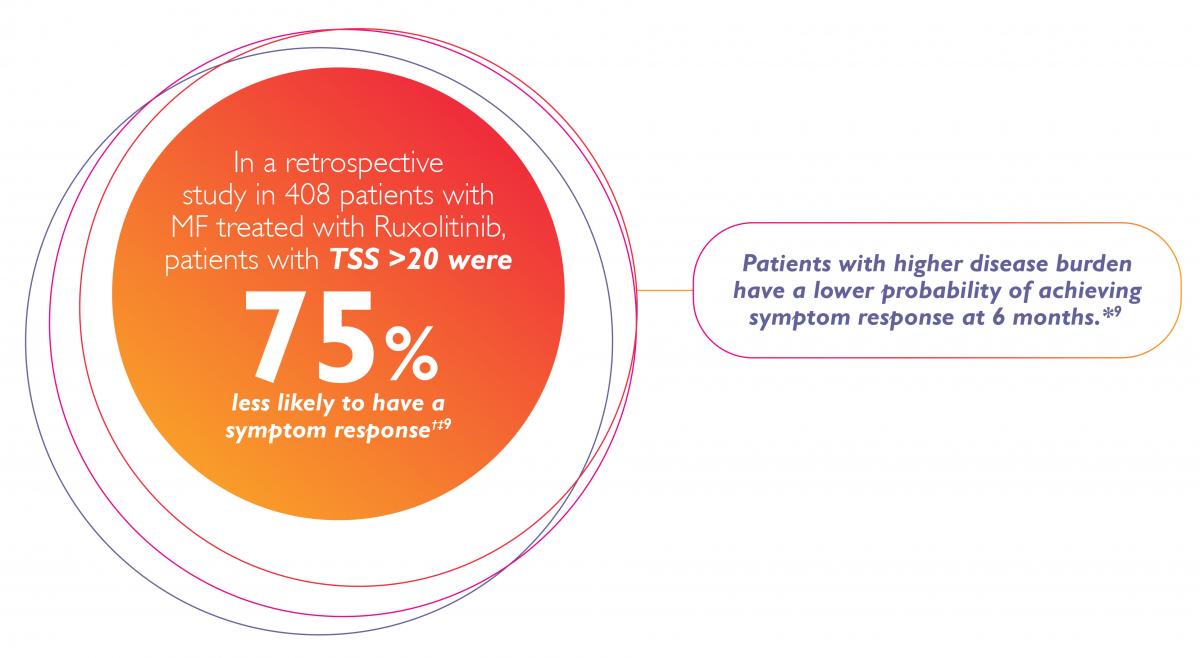
Proactively monitor all your patients’ MF-related symptoms to treat as early as possible those who can benefit from Ruxolitinib (JAKAVI).1–4
JUMP was a Phase 3b expanded access trial (N=2233) assessing the efficacy and safety of Ruxolitinib (JAKAVI) in patients with intermediate- or high-risk MF (n=1114), intermediate-1-risk MF (n=163) and low-risk MF (n=60). Primary endpoint was assessment of Ruxolitinib (JAKAVI) tolerability by the frequency, duration and severity of AEs.10,11
* Data from a retrospective analysis of 408 patients with MF treated with Ruxolitinib (JAKAVI).9
† OR=0.25, 95% CI (0.13–0.47), p<0.001.9
‡ A symptom response is defined as a ≥50% reduction in the Myeloproliferative Neoplasm Symptom Assessment Form Total Symptom Score (MPN-SAF TSS).9
§ Data from an ad-hoc analysis of the safety and efficacy of Ruxolitinib (JAKAVI) in a subset of patients in JUMP with DIPSS low-risk MF (n=60).10
Abbreviations
AE, adverse event; CI, confidence interval; DIPSS, dynamic international prognostic scoring system; JUMP, JAK Inhibitor Ruxolitinib in Myelofibrosis Patients; MF, myelofibrosis; OR, odds ratio; QoL, quality of life; TSS, Total Symptom Score
References:
-
Reilly J, et al. Br J Haematol. 2014;167:418–438.
-
National Institute for Health and Care Excellence. Ruxolitinib for treating disease-related splenomegaly or symptoms in adults with myelofibrosis [TA386]. 2016. Available at: https://www.nice.org.uk/guidance/ta386. Last accessed April 2020.
-
Novartis Healthcare Phils., Inc. (2019 September). Ruxolitinib (Jakavi) Tablets; Prescribing Information. Makati, Philippines.
-
Mesa R, et al. Blood. 2012;120:Abstract 1727.
-
Mesa R, et al. BMC Cancer. 2016;16:167.
-
Vannucchi A, et al. Haematologica. 2015;100:1139–1145.
-
Novartis Healthcare Phils., Inc. (2018 April). Ruxolitinib (Jakavi) Tablets; Basic Succinct Statement. Makati, Philippines.
-
Vannuchi A and Harrison C. Blood. 2017;129:693–703.
-
Palandri F, et al. Oncotarget. 2017;8:79073–79086.
-
Guglielmelli P, et al. EHA 2018, 14–17 June; Stockholm, Sweden. Poster PF623.
-
Al-Ali H, et al. Haematologica. 2016;101:1065–1073.

